Accordingly, you may need to go beyond your checklist to dig deeper and That being said, you are encouraged to explore problems to the fullest extent possible rather than skipping over a problem so you can touch lightly on other subjects. It is the auditor’s job to keep the auditee on track and extract the information needed. Don’t go down the rabbit hole with someone who is trying to explain something that is irrelevant. Be polite but insist on getting details needed to answer the question.

In these cases, it is important that you remain courteous but persistent. You should dictate the documents you want to see, reviewing the requisite number of samples stipulated in your audit plan.ĭuring the audit, you will invariably come across people who nervously ramble, digress, or are intentionally vague or evasive. You need to be sure that the auditee is not cherry-picking documents to show you. Consider stratified random sampling to focus the audit based on risk (e.g., rather than taking a random sampling of purchase orders, stratify the population by criticality to focus on what is important).
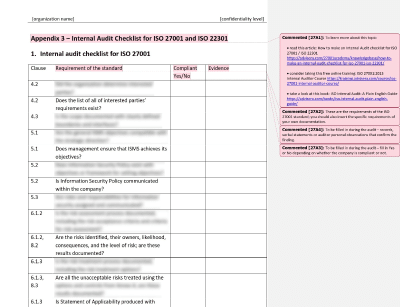
When you start performing the audit, it is important to remember that an audit is really a method of sampling and is conducted to get a sense of what is happening. People – not documents – make or break a system. You should make every effort to deal directly with the people involved in implementing the system. Conducting the On-Site Audit and Avoiding Rabbit HolesĪll that preparation you did in the weeks leading up to the audit will now pay off.
#Iso 13485 internal audit checklist iso
These types of audits can be considerably longer than an ISO audit. MDSAP audits are based on the number of elements to be covered in the audit. It should be noted, however, that this type of audit length determination is trending out with the use of audit duration calculations used in the Medical Device Single Audit Program Model (MDSAP). The International Accreditation Forum documents MD-5 and MD-9 set guidelines for internal audit days as well as general protocols for conducting an ISO audit.
#Iso 13485 internal audit checklist manual
For example, there is certainly more risk associated with manufacturing heart valves than manual wheelchairs, and this impacts audit length. The risk associated with the device is also a factor. ISO audit duration is based on the number of employees in the facility and the scope of the QMS. Confirm the time of the closing meeting, and state how long it will take after that meeting until the audit report is issued.Confirm the working hours, meal breaks, and time for daily debriefings.State the audit objectives and emphasize that the audit will try not to interfere with operations.notes, recording findings, classifying nonconformities, etc.).Describe the audit methodology (e.g., interviewing, observing, reviewing documentation, taking.Confirm the purpose and scope of the audit, and confirm the audit plan (typically sent a few weeks.Discuss the responsibilities of auditee management.

